 If an electron neutrino is emitted, as in #p + e⁻ → n + ν_e#, the sums of the lepton numbers are +1 on the left and +1 on the right. This process reduces the atomic number by 1 and emits gamma radiation or an x-ray and a neutrino. If the process were simply #p + e⁻ → n#, the sum of the lepton numbers would be +1 on the left and 0 on the right. Electron capture is a type of radioactive decay where the nucleus of an atom absorbs a K or L shell electron and converts a proton into a neutron. A very introductory level description of electron capture (for a general chemistry audience)Be sure to visit my site for lessons with embedded qu. 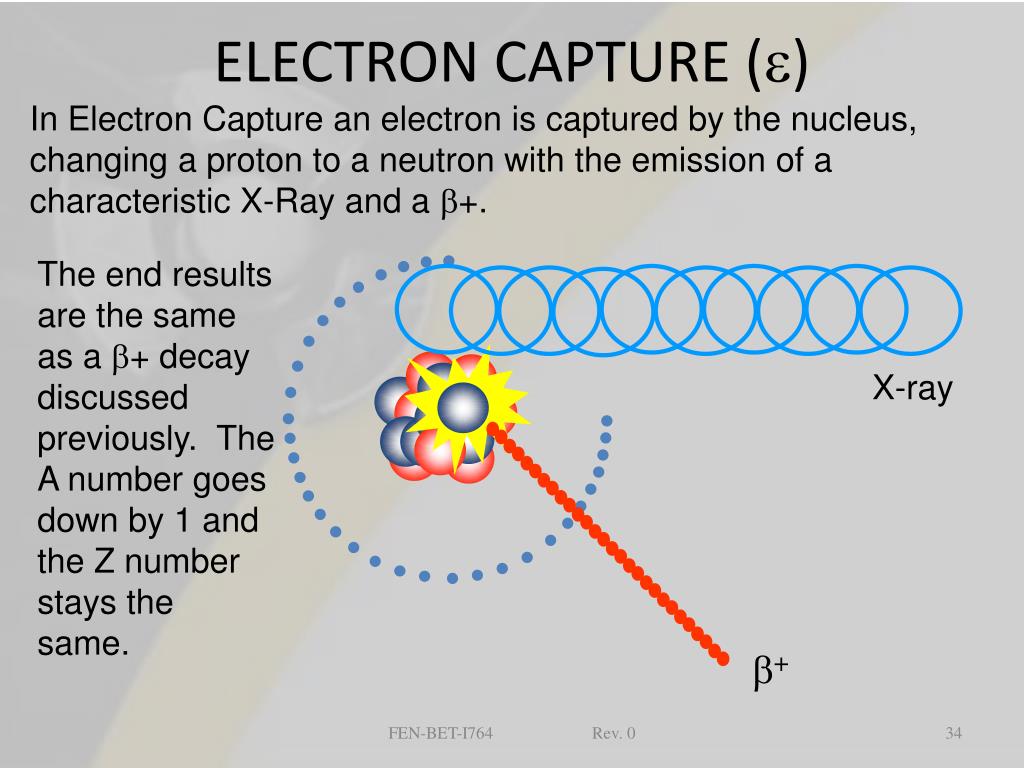
A Novel Use of Negative Ion Mobility Spectrometry for Measuring Electron Attachment Rates. Protons and neutrons are held together in the nucleus by the strong nuclear force. This article is cited by 50 publications. The lepton numbers of electrons and of electron neutrinos are +1. The two most common modes of natural radioactivity are alpha decay and beta decay. Protons and neutrons are made up of quarks. 
The benzene-like structure is found to be the most. This process reduces the atomic number by 1 and. Charge distribution and nucleus independent chemical shift (NICS). > electron capture - Electron capture is a type of radioactive decay where the nucleus of an atom absorbs a K or L shell electron and converts a proton into a neutron. The electron converts a proton to a neutron in the nucleus. The atomic numbers and mass numbers in a nuclear equation must be balanced. electron capture: capture of an electron by an unstable nucleus. Electrons and neutrinos have spin ½ and are outside the nucleus, so they are leptons. A nuclear reaction is one that changes the structure of the nucleus of an atom. 
One of the rules that must be followed in nuclear reactions is the Law of Conservation of Lepton Number.Ī lepton is an elementary particle with spin ½ that does not undergo strong nuclear forces. You are probably familiar with conservation laws like the Law of Conservation of Energy and the Law of Conservation of Momentum. Correlations between molecular structure and fragmentation observed in electron capture negative chemical ionization mass spectra (moderator gas methane). In general, the purpose of an electron capture detector is to detect the concentration of the analyte based on how many electrons it absorbs to use in an electron capture event. Electron capture produces an electron neutrino in order to conserve lepton numbers.Įlectron capture is a process in which one of the inner electrons of an atom is captured by a proton in the nucleus, forming a neutron and emitting an electron neutrino. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
